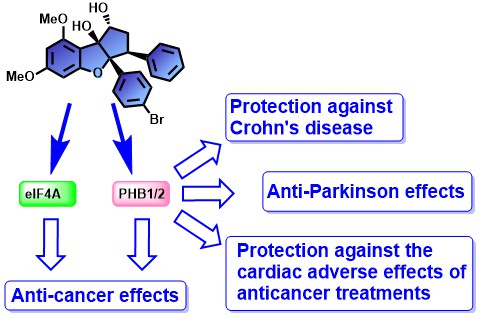
Flavaglines represents a family of plant natural products used in Traditional Chinese Medicine that display a potent cytotoxicity in cancer cells and cytoprotective effect in non-cancer cells. Our lab was the first to develop synthetic flavaglines that display enhanced pharmacological properties compared to natural flavaglines. We also developed some chemical tools that were used to identify prohibitins-1 and 2 (PHB1/2) as the molecular targets of flavaglines. These two scaffold proteins regulate various cellular functions, including proliferation, survival, tumor suppression, transcription and mitophagy. Flavaglines also target another protein: the translation initiation factor eIF4a.
We demonstrated that in addition to their antitumor activities, flavaglines protect the heart against the cardiac adverse effects of anticancer treatments. They also protect the colon in a mouse model chronic inflammatory bowel disease (Crohn’s disease).
Overall, our work suggests that flavaglines could improve the efficacy of chemotherapies and alleviate in the same time their adverse effects, in particular cardiotoxicity.
With our collaborators, we demonstrated that flavaglines target prohibitins, which are scaffold proteins that modulate many signaling pathways controlling cell survival, metabolism and inflammation. These compounds inhibit also another protein: the translation initiation factor eIF4a.
With our collaborators in Europe, America and Asia, we are currently pursuing the following objectives:
- to complete the characterization of the Structure-Activity Relationships (SAR) of several classes of prohibitin ligands for their various pharmacological effects,
- to synthesize chemical tools to decipher in detail the action of prohibitin ligands in different physiological and pathological conditions,
- to define the therapeutic potential of flavaglines and other prohibitin ligands against cancers, cardiovascular, inflammatory, metabolic and neurological diseases,
- to develop other classes of anticancer agents unrelated to prohibitin ligands with original mode of action.